Inositol pyrophosphate IP7
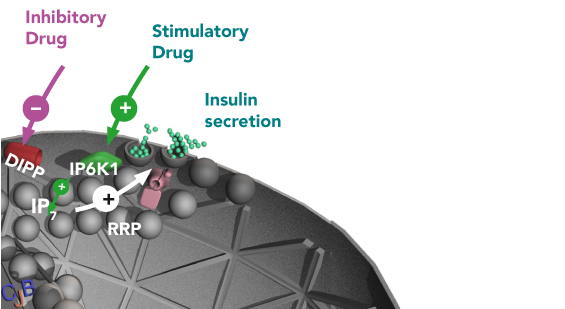
High amounts of IP7 are necessary for efficient insulin release (green cicles) by beta-cells. IP7 stimulates the ready releasable pool (RRP) of insulin to be secreted to the blood. IP6K1 builds IP7 from IP6, while DIPP reverses this action. To increase the amounts of IP7 in diabetic patients, DIPP should be blocked or IP6K1 should be stimulated with a specifically designed inhibitory or stimulatory drug, respectively.
.
.
.
.
.
Inositides contribute to vital cell processes
Inositides take an active part in regulating many vital processes in all cells in the organism. Depending on its primary function, every cell has a specific footprint of what inositides it contains.
Beta-cells contain unusually high amounts of the inositol pyrophosphate IP7
One of the inositides is an inositol pyrophosphate with a common name IP7. It is an active molecule and as soon as it is formed with the assistance of the IP6K1 enzyme, it is degraded back to the more stable IP6 under the control of DIPP enzyme. In insulin producing beta-cells the amount of IP7 is unusually high compared to other cells in mammals. IP7 in beta-cells controls insulin release.
For efficient glucose metabolism insulin is released in two phases
As levels of blood glucose rise, it is taken up by beta-cells. This initiates a series of molecular events leading to insulin being released into the blood. Insulin is released in two phases, an initial and a second phase. This enables a more efficient regulation of blood glucose levels.
High levels of IP7 in beta-cells guarantee efficient insulin release
We have shown that IP7 increases the size of the pool of insulin granules that are available for the first phase of secretion. This is known as the readily-releasable pool. Therefore maintaining a high level of IP7 in beta cells is vital for an initial quick and efficient insulin release and the proper control of blood glucose concentration.
Increasing levels of IP7 could be a cure for type 2 diabetes patients
Patients with type 2 diabetes have an impaired first phase insulin release compared to healthy individuals. One way to promote first phase insulin release might be to prevent degradation of IP7 through reduction of DIPP activity. Another way is to make more IP7 by enhancing IP6K1.
.
.
.
.
Read more about IP7:
- Inositol pyrophosphates and Akt/PKB: Is the pancreatic β-cell the exception to the rule? Kim J et al. Cell Signal. 2019 Jun;58:131-136.
- Inositol hexakisphosphate kinase 1 is a metabolic sensor in pancreatic β-cells. Rajasekaran SS et al. Cell Signal. 2018 Jun;46:120-128.
- Protein kinase- and lipase inhibitors of inositide metabolism deplete IP7 indirectly in pancreatic β-cells: Off-target effects on cellular bioenergetics and direct effects on IP6K activity. Rajasekaran SS et al. Cell Signal. 2018 Jan;42:127-133.
- New horizons in cellular regulation by inositol polyphosphates: insights from the pancreatic β-cell. Barker CJ, Berggren PO. Pharmacol Rev. 2013 Feb 19;65(2):641-69
- The pancreatic beta cell as a pardigm for advances in inositide research. Barker CJ, Berggren PO. Advances in Biological Regulation. 2012 Sep;52(3):361-8
- Inositol hexakisphosphate suppresses excitatory neurotransmission via synaptotagmin-1 C2B domain in the hipppocampal neuron. Yang SN, Shi Y, Yang G, Li Y, Yu L, Shin OH, Bacaj T, Südhof TC, Yu J, Berggren PO. Proc Natl Acad Sci U S A. 2012 Jul 24;109(30):12183-8.
- Diphosphoinositol pentakisphosphate as a novel mediator of insulin exocytosis. Barker CJ, et al., Adv Enzyme Regul.2009;49(1):168-73.
- Requirement of inositol pyrophosphates for full exocytotic capacity in pancreatic beta cells. Illies C, et al., Science. 2007 Nov 23;318(5854):1299-302.

